Evidence of Work:
Our task for this project was to create four pieces of art for the new STEM building. Because it is for the STEM building, we were required to use and explain the chemistry that was happening when creating the art piece. We had to have an overall theme or issue that we wanted to address with the four pieces of art. We started off with the idea to create something using a combustion reaction with gunpowder on wood. After creating a gunpowder-like compound, we added fire and it didn't burn the way we wanted it to so we had to come up with another idea. Our second attempt was using ammonium chloride and a heat gun to burn the wood. This was a decomposition reaction because the ammonium chloride was broken into two different compounds of ammonia gas and hydrochloric acid. The reaction is NH4CL - NH3 + HCl. To create this reaction we used small paint brushes to paint the clear ammonium chloride onto the untreated wood. After, we went over the painted places with the heat gun to help the reaction occur. The ammonia gas was released into the atmosphere and the hydrochloric acid was left behind, burning the wood. Our collection was put together in a similar formation as it is below with our place card in the middle. The place card had all the information about the message and the chemical reactions.
Our task for this project was to create four pieces of art for the new STEM building. Because it is for the STEM building, we were required to use and explain the chemistry that was happening when creating the art piece. We had to have an overall theme or issue that we wanted to address with the four pieces of art. We started off with the idea to create something using a combustion reaction with gunpowder on wood. After creating a gunpowder-like compound, we added fire and it didn't burn the way we wanted it to so we had to come up with another idea. Our second attempt was using ammonium chloride and a heat gun to burn the wood. This was a decomposition reaction because the ammonium chloride was broken into two different compounds of ammonia gas and hydrochloric acid. The reaction is NH4CL - NH3 + HCl. To create this reaction we used small paint brushes to paint the clear ammonium chloride onto the untreated wood. After, we went over the painted places with the heat gun to help the reaction occur. The ammonia gas was released into the atmosphere and the hydrochloric acid was left behind, burning the wood. Our collection was put together in a similar formation as it is below with our place card in the middle. The place card had all the information about the message and the chemical reactions.
|
Our Message: As a group we wanted to bring awareness to the dangers of pollution and human damage to the earth. We had four panels of wood that showed the dangers of different kinds of pollution. The first one was the cut down tree representing deforestation and the harms that come with it. The next one was of trash in the ocean to show the harm humans are doing to ocean ecosystems. The third one is a busy city to put emphasis on air pollution and the thing we are doing to harm the earth. Lastly, to bring everything together we created an earth on fire. We are causing a lot of harm to the environment and not really caring or doing anything about it. In the future our world could be destroyed because of these things we are doing now.
|
Content:
Chemical Reaction- one or more substances react and change into new substances. The new compounds that have been formed have different properties then the reactants. The amount of matter has to be the same on both sides of the reaction to satisfy the law of conservation of mass.
Balancing Chemical Reactions- for a reaction to be balanced, there has to be the same amount of mass on both sides. For example, the reaction H2+O2 - H2O is not balanced because there are two oxygen on the reactant side and only one on the product side. To balance this equation you would need to double the hydrogen and water to make it equal mass on both sides. The final equation would look like 2H2+O2 - 2H2O. There are now four hydrogen and two oxygen on the reactant side and four hydrogen and two oxygen on the product side.
Synthesis Reaction- a chemical reaction where two compounds react to form one completely new compound. An example of this would be A+B - AB.
Decomposition Reaction- a chemical reaction where one compound breaks down into two different compounds. An example of this would be AB - A+B.
Single Replacement Reaction- a reaction where one element will replace another element that is in an alike ionic compound. This means that a single element will kick out another element in compound if the outside element is more reactive. In this type of reaction the product normally ends up as a single element and a compound. To know if the reaction will work the element by itself has to be higher on the reactivity series then the one in compound. An example of this could be A+BC - AC+B.
Double Displacement Reaction- a reaction where two compounds switch ions to create two new compounds. This reaction normally produces a gas, liquid, or precipitate. The solubility guidelines have rules that will tell you what will be formed when two elements are reacting with one another. An example of this is AB+CD - AD+CB.
Combustion Reaction- a reaction where carbon dioxide and water are produced due to some kind of combustion. This normally requires some type of heat to set off the chemical equation. An example of this is any hydrocarbon+O2 - CO2+H2O.
|
Solubility Guidelines- these guidelines can tell us what will be formed in different chemical reactions depending on the ionic compounds. If it is one of the soluble compounds then it will be a liquid. But if it is one of the exceptions then it will be insoluble. It's the same with the insoluble compounds. If it contains one of those compounds then a precipitate will be formed. But the exceptions mean it will be soluble and it will only be a liquid.
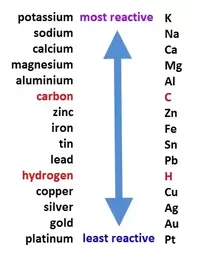
Reactivity Series- this guideline helps with single replacement reactions because it is a list of metals in order of reactivity. The ones closer to the top are more reactive then the ones near the bottom. If a single replacement reaction were to occur, the single metal has to be higher on the list to replace the one in compound. If it isn't then no reaction would take place. |
Reflection:
This project was a little difficult but we managed to push through and finish the project. I really liked how everything turned out even though it wasn't what we originally wanted. As a group we did a really good job at persevering and being open minded. We had about a week and a half to complete this project and we spent most of the first week trying out the gunpowder idea. We knew we had to change our project so we worked hard to finish the project on time. While doing this we collaborated really well. Each person was assigned a board and we all worked on different sections of the project like the slideshow, place card, and the research. Another thing we did well was being open minded. When the first attempt didn't work, we tried it again with a few changes but the gunpowder-like compound didn't burn the way we wanted it to. Then we researched other ways to burn the wood with a chemical reaction and came across the ammonium chloride. We didn't have much time so we tried it and it worked really well. Being open minded allowed us to finish the project with a good product. While this project showed that we could preserver, it also showed me what I need to work on for the next project. I made progress on becoming a better leader but I still need to work on it. When we would not know what to do next I would sometimes speak up but other times I stayed quiet and waited for someone else to say an idea. Another thing I need to work on for next time is critical thinking. When we had a problem we asked for help right away before taking time to think about a possible solution. I could have researched the problem and came up with a solution that way. This project was really fun because it brought together chemistry and art. For next time I want to continue progressing my leadership skills and thinking more before asking for help.
This project was a little difficult but we managed to push through and finish the project. I really liked how everything turned out even though it wasn't what we originally wanted. As a group we did a really good job at persevering and being open minded. We had about a week and a half to complete this project and we spent most of the first week trying out the gunpowder idea. We knew we had to change our project so we worked hard to finish the project on time. While doing this we collaborated really well. Each person was assigned a board and we all worked on different sections of the project like the slideshow, place card, and the research. Another thing we did well was being open minded. When the first attempt didn't work, we tried it again with a few changes but the gunpowder-like compound didn't burn the way we wanted it to. Then we researched other ways to burn the wood with a chemical reaction and came across the ammonium chloride. We didn't have much time so we tried it and it worked really well. Being open minded allowed us to finish the project with a good product. While this project showed that we could preserver, it also showed me what I need to work on for the next project. I made progress on becoming a better leader but I still need to work on it. When we would not know what to do next I would sometimes speak up but other times I stayed quiet and waited for someone else to say an idea. Another thing I need to work on for next time is critical thinking. When we had a problem we asked for help right away before taking time to think about a possible solution. I could have researched the problem and came up with a solution that way. This project was really fun because it brought together chemistry and art. For next time I want to continue progressing my leadership skills and thinking more before asking for help.